 Immobilization also increases stability of the enzyme against heat denaturation and autolysis and results in longer maintenance of activity. These adducts can be detrimental to fragments in the presence of reductants. 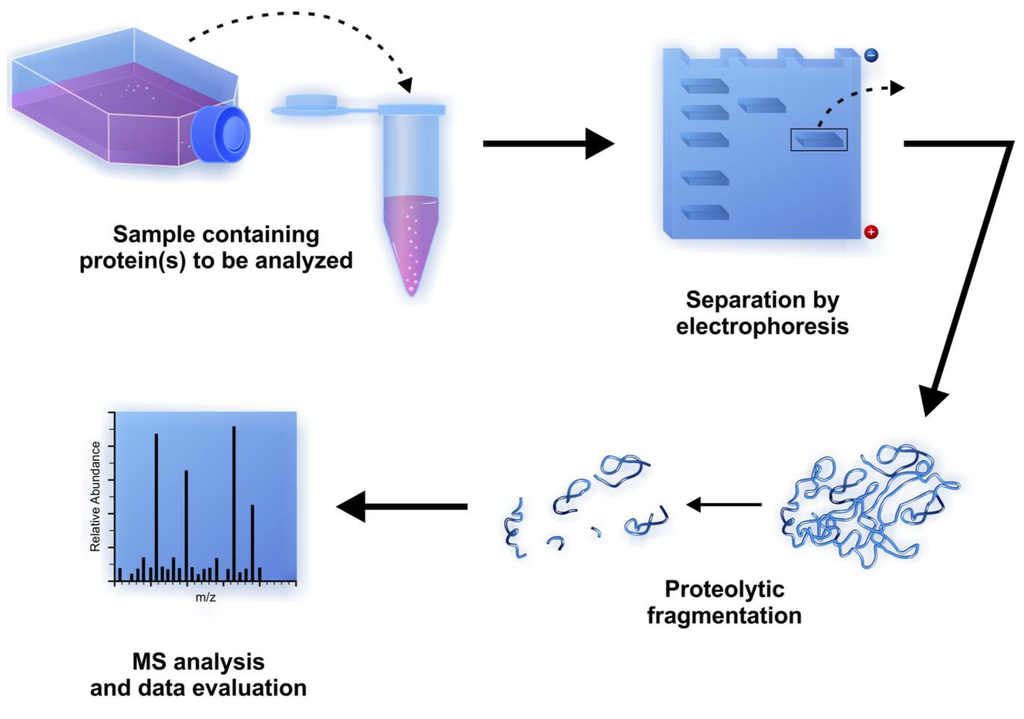
The use of immobilized papain alsos prevent formation of antibody-enzyme adducts, which can occur when using the soluble form of sulfhydryl proteases (such as papain). Thus, there is no need to develop an ion exchange method for separating the fragments from the enzyme. Immobilized papain (i.e., Papain Agarose Resin) is the preferred reagent because it allows for easy control of the digestion reaction and quick removal of enzyme from the digestion products following incubation. Mercuripapain, which is less prone to autodigestion than crystalline papain, can be used however, both of these non-immobilized enzymes require an oxidant to terminate digestion. If the cysteine is not completely removed, over digestion can be a problem (2).Ĭrystalline papain is often used for the digestion of IgG however, it is prone to autodigestion. These fragments are often inconsistent, and reproducibility can be a problem. If no cysteine is present during papain digestion, F(ab')2 fragments can be generated. The excess cysteine is then removed by gel filtration. To prepare F(ab')2 fragments, the papain is first activated with 10mM cysteine. Papain is primarily used to generate Fab fragments, but it also can be used to generate F(ab')2 fragments (2). Learn more: Tech Tip #59: Secondary antibodies: A guide to fragment specificity Lower immunogenicity than intact antibody for experiments in vivo.Simpler system for studying the structural basis for immune recognition using X-ray crystallography or NMR.complement fixation) in antigen-antibody binding studies Elimination of Fc-associated effector functions (e.g.Potentially higher sensitivity in antigen detection in solid phase applications as a result of reduced steric hindrance from large protein epitopes.More efficient penetration of tissue sections, resulting in improved staining in immunohistochemistry (IHC).Ability to control Fc-binding to Protein A or Protein G in experiments involving immunoprecipitation and Western blotting.Reduced nonspecific binding from Fc interactions (many cells have receptors that bind the Fc region).For these reasons, fragmentation is usually performed only when the antibody of interest is available in large quantity and the particular application demands it.īecause of their smaller size as functional components of the whole molecule, antibody fragments offer several advantages over intact antibodies for use in certain immunochemical techniques and experimental applications: Fc fragments consist of the heavy chain constant region (Fc region) of an immunoglobulin and mediate cellular effector functions.Īntibody fragmentation is somewhat laborious, requires optimization of enzyme-mediated digestion of the protein, and necessitates an ample supply of antibody (e.g., 10mg) to make it reasonably efficient. Several types of antigen-binding fragments are possible, but each contains at least the variable regions of both heavy and light immunoglobulin chains (VH and VL, respectively) held together (usually by disulfide bonds) so as to preserve the antibody-binding site. 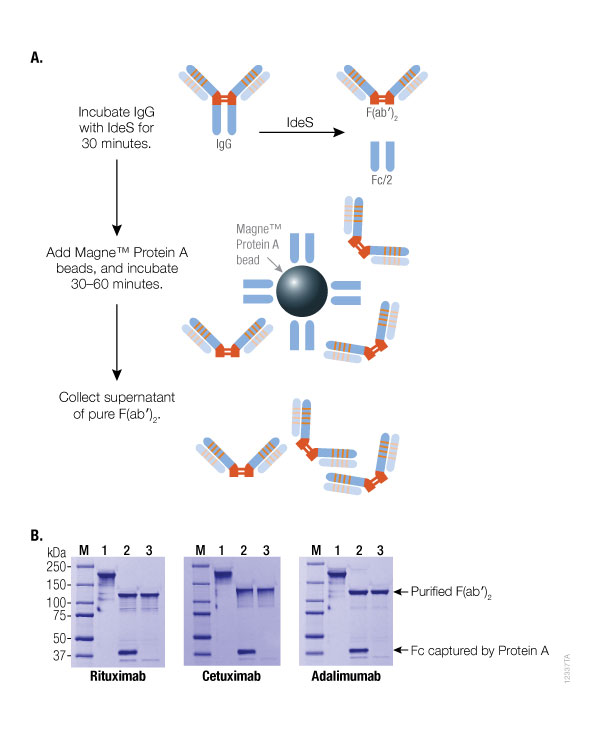
The two groups of antibody fragments of primary interest are (a) antigen-binding fragments such as Fab and (b) class-defining fragments such as Fc that do not bind antigen. Although fragmentation of all immunoglobulin classes is possible, only procedures for fragmentation of mouse, rabbit, and human IgG and IgM have been well characterized. Antibody fragmentation is accomplished using reducing agents and proteases that digest or cleave certain portions of the immunoglobulin protein structure. It is possible to selectively cleave the immunoglobulin molecule into fragments that have discrete characteristics. Sometimes it is useful to study or make use of the activity of one portion of an immunoglobulin without interference from other portions of the molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
